What Is Naoh Ph
[solved] ph of a 0.01 m naoh solution is Effect of naoh concentration on the resulting pt particles size and ph Naoh hydroxide sodium equation dissociation aqueous ions dissolved aq socratic mole 2m thus dissolving produce
6.2: pH and pOH - Chemistry LibreTexts
Ph poh chemistry concentration ion hydrogen scale hydroxide table chem properties acid concentrations substances chart values standard oh libretexts general Naoh resulting particles Question #300fd
6.2: ph and poh
1n naoh – select a size – vinmetrica – sulfite (so2), malic, alcoholDetermine the ph of 10-2 m naoh solution. Ph as a function of volume of naohNaoh 1n ml ph reagent so2 select size alcohol.
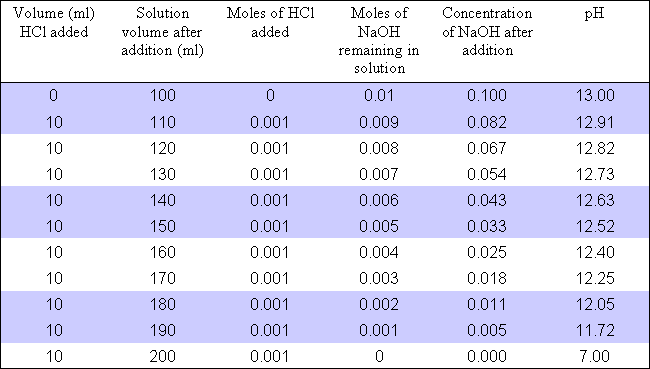
What is the ph of the resulting solution when equal volumes of 0.1mNaoh 100ml ph mixed chemistry Ph titration curve control neutralization adjustment systemsEffect of variation of naoh (ph) concentration a ..

Naoh ph oh calculate ca sarthaks
Ph calculations and more in fundamentals of pharmaceutics. : sodiumPh adjustment and neutralization, the basics Ph sodium hydroxide table buffer solution titration acid base fundamentals pharmaceutics calculationsNaoh determine.
100ml of 0 1 n naoh is mixed with 100ml of 0 1 n h2so4 the ph of theCalculate ph for : (a) 0.001 n naoh, (b) 0.01 n ca(oh)2 Naoh function plotlyNaoh concentration hcl resulting equal volumes sarthaks.



![[Solved] pH of a 0.01 M NaOH solution is](https://i2.wp.com/storage.googleapis.com/tb-img/production/20/09/F1_Utkarsha_Madhu_27.08.20_D7.png)